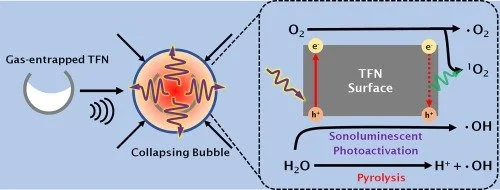
Sonochemical Reaction Engineering
Sonochemistry is the use of ultrasound and cavitation to promote chemical reactions or facilitate nanoparticle formation. Our interests here are to engineer and study novel sonochemical reactors and sonocatalysts to promote novel chemistries. We are particularly keen on exploring green hydrogen, ammonia chemistry, CO2RR, and AOP to name a few.
Relevant Papers
“Unsupported gold nanocones as sonocatalytic agents with enhanced catalytic properties”, Ultrasonics Sonochemistry
“Nanostructured TiO2 cavitation agents for dual-modal sonophotocatalysis with pulsed ultrasound”, Ultrasonics Sonochemistry
”Sol–Gel Transitions of Comb‐Like Polymethacrylate Copolymers by Mechano‐Thermal Stimuli in Water”, Macromolecular Chemistry and Physics
Ultrasound Enhanced Therapies
A number of diseases remain difficult to treat due to the inability to reliably deliver potent therapeutics to the site of the injury. Our group is interested in designing and developing novel ultrasound devices and cavitation nuclei to enhance the treatment to diseases such as cancer and peripheral artery disease. For example, we continue to develop novel ultrasound-responsive drug-loaded micro- and nanoparticles that nucleate cavitation. Our current focus is to develop novel methods to prevent restenosis post angioplasty. We currently are developing drug-eluting microparticles designed to implant into the arterial lesion in response to ultrasound, whereby they will slowly release the drug.
Relevant Papers
“Ultrasonic Implantation and Imaging of Sound-Sensitive Theranostic Agents for the Treatment of Arterial Inflammation”, ACS Applied Materials & Interfaces
“Investigating the Acoustic Response and Contrast Enhancement of Drug-Loadable PLGA Microparticles with Various Shapes and Morphologies”, Ultrasound in Medicine & Biology
”Remote targeted implantation of sound-sensitive biodegradable multi-cavity microparticles with focused ultrasound”, Scientific Reports
“Biomedical applications for Gas-Stabilizing Solid Cavitation Agents”, Langmuir